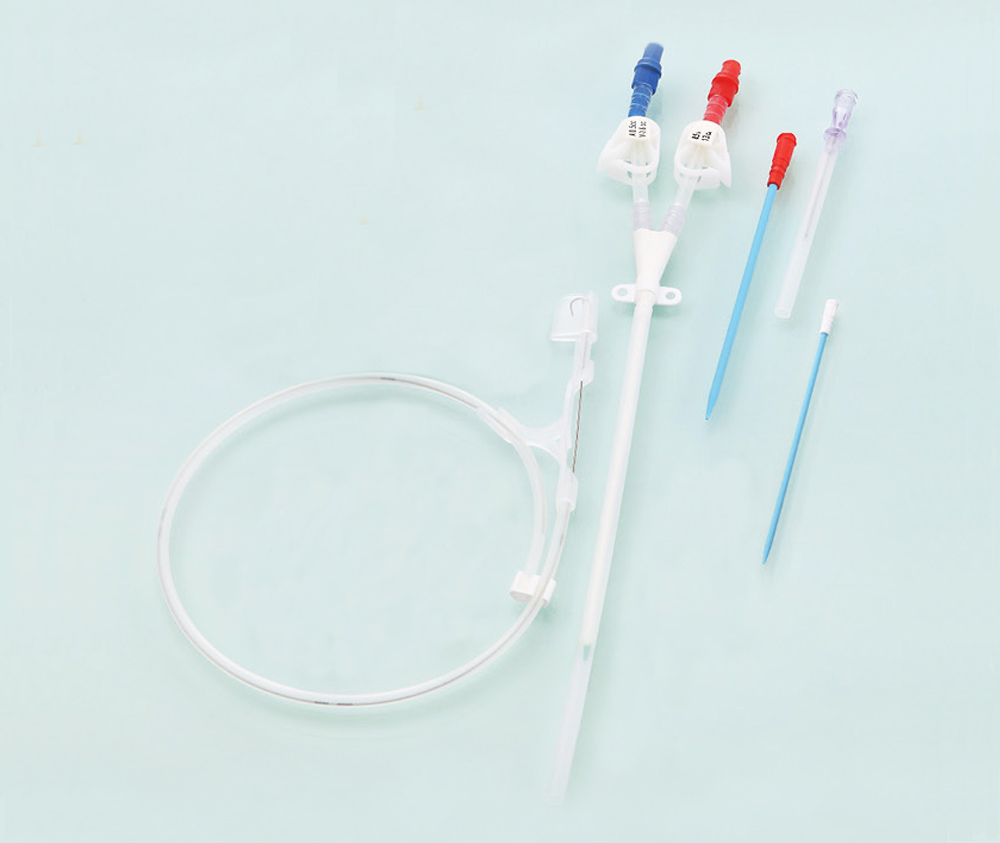
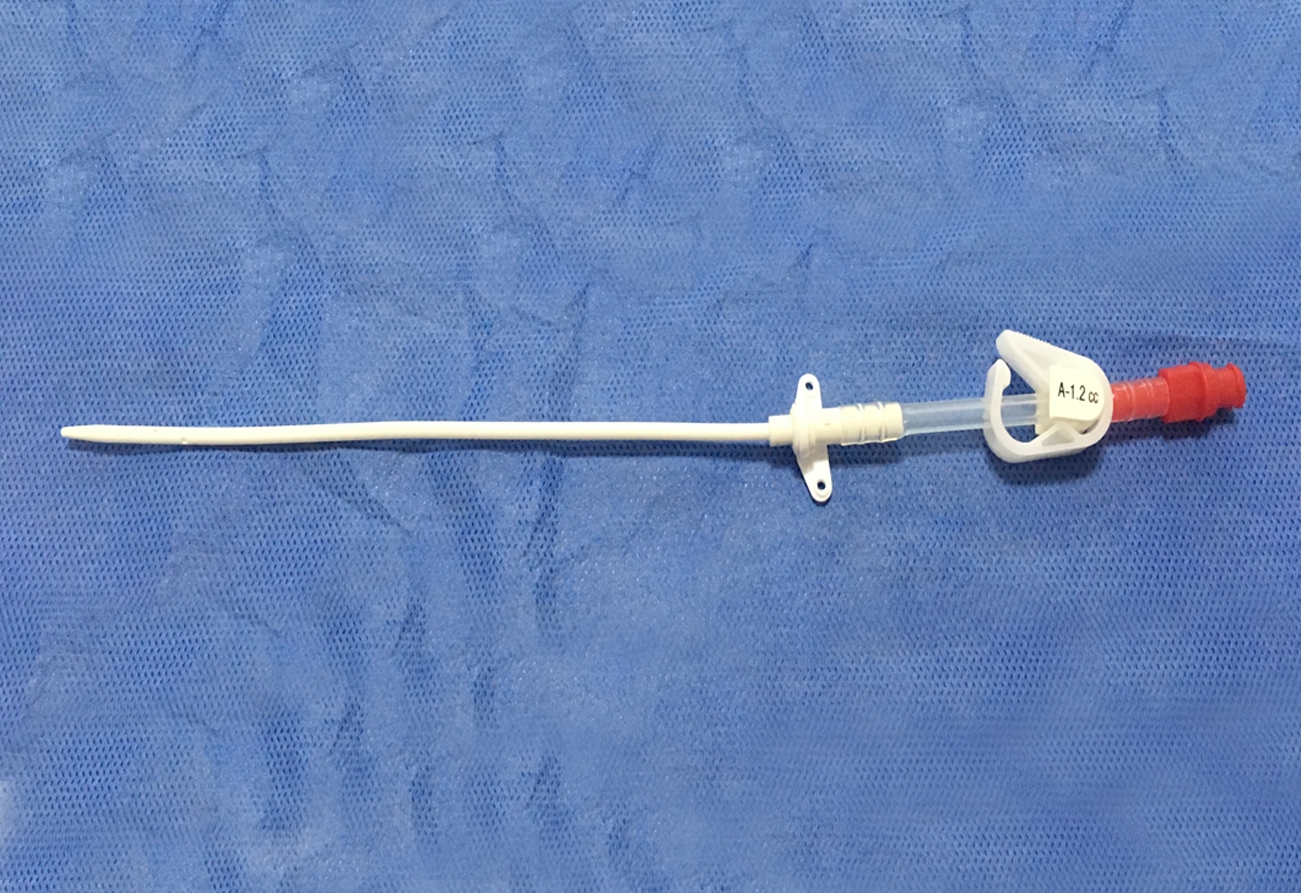
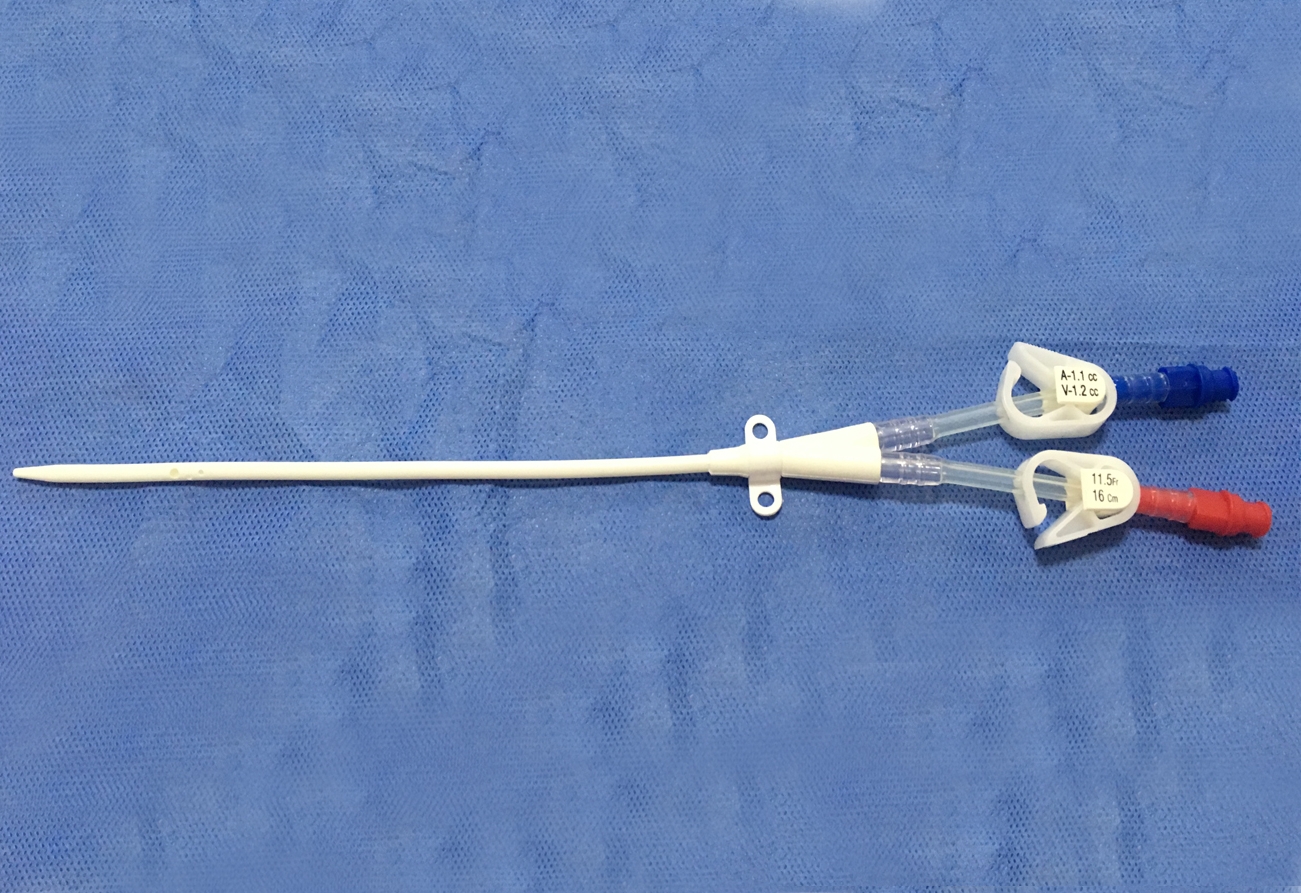
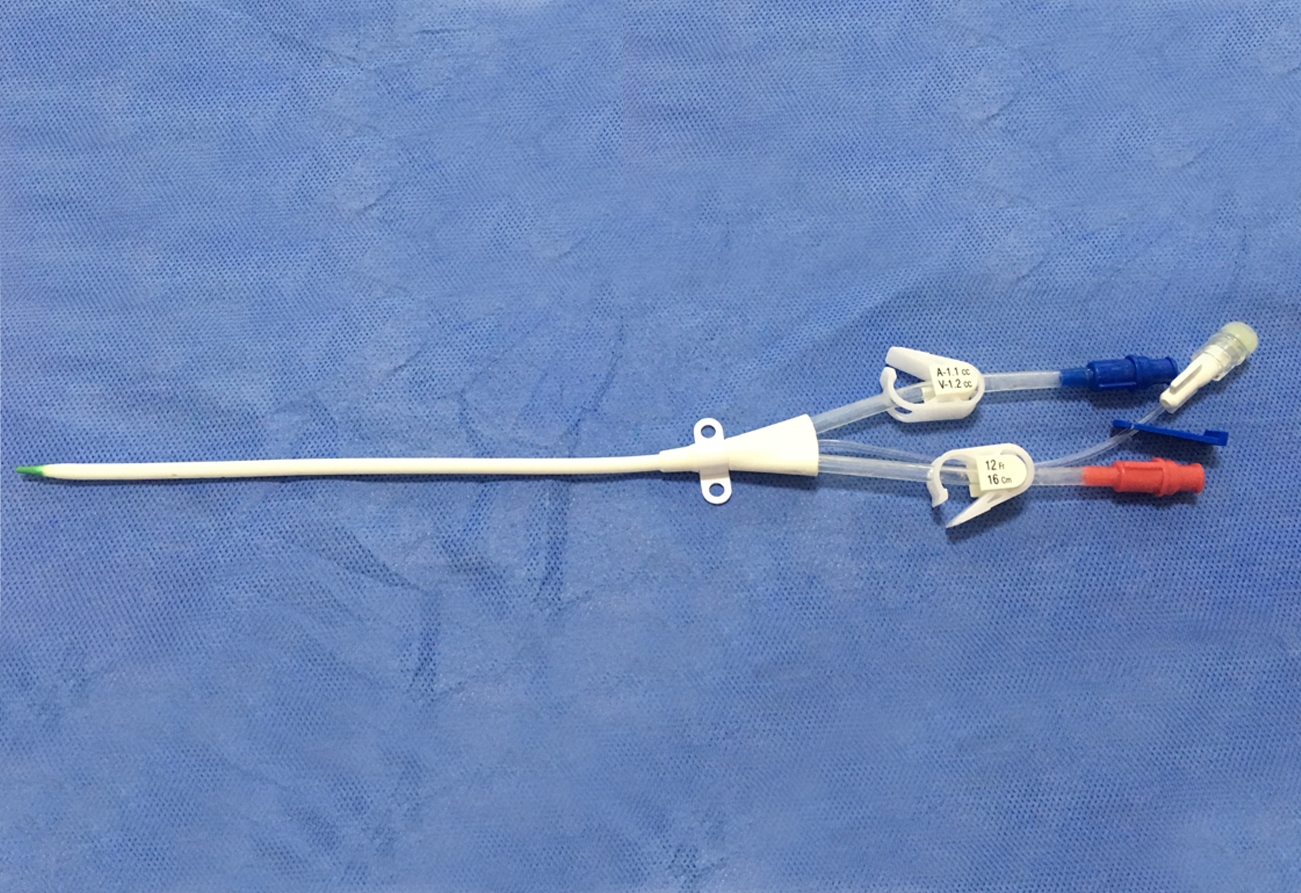
The Haemodialysis Catheter (HC) is usually placed in deep vein, like the neck (jugular vein) or chest (subclavian vein) via seldinger technique to build a temporary blood connection between the patient and the dialysis machine for acute and short-term dialysis. The advantages for this technique are: small scar, convenient to use, suitable for short-term implantation and large blood flow, etc., thus it has widely been used in clinical treatment.
The catheter tube in Kit is made of medical grade polyurethane and silicone rubber. The components and accessories ensure the product with safety and feasibility. The kit includes puncture needle, dilator, and guidewire etc., which makes the operation easier as follows: X-ray opaque; Conical elastic soft tip to protect vessel inner wall; Polyurethane with elasticity and flexibility; Large smooth lumen design with enhanced flux and vacuum resistance; Transparent extension tube, made from silicone rubber, makes the flow condition visible; Scaled J-tip guidewire to protect vessel inner wall, and facilitate in knowing the insertion depth of guidewire; Infection preventive heparin injection cap.
Intended Use and Indication
Warning and Precautions
Contraindications and Complications
Order Information
Advertising Review Filing Number
SSCP
Intended Purpose
The Haemodialysis Catheter Kit is intended for building a temporary
blood connection between the patient and the dialysis machine. The flexible
tubing permits percutaneous insertion into subclavian, jugular, and femoral
veins.
- The Double Lumen Haemodialysis Catheter is intended for short-term central venous access for haemodialysis, infusion, and pressure monitoring.
- The Triple Lumen Haemodialysis Catheter is intended for short-term central venous access for haemodialysis, infusion, and pressure monitoring.
Indications
- Acute renal failure due to various causes;
- End-stage renal disease of various causes;
- Drug overdose or food poisoning;
- Severe water retention, such as nephrotic syndrome, chronic cardiac insufficiency, liver cirrhosis, etc;
- Electrolyte disturbances, such as hyperkalemia, hypermagnesemia, etc;
- Other, such as liver coma, psoriasis, hyperuricemia and so on.
The information is sourced from the Instruction for Use (IFU-01-02, version number: 2026A).
1. Sterile, Single use: Do not reuse, reprocess or resterilize. Reuse of device creates a potential risk of serious injury and/or infection which may lead to death. Reprocessing of medical devices intended for single use only may result in degraded performance or a loss of functionality.
2. Read all package insert warnings, precautions and instructions prior to use. Failure to do so may result in severe patient injury or death.
3. Do not place/advance catheter into or allow it to remain in the right atrium or right ventricle. The catheter tip should be advanced into the lower 1/3 of the Superior Vena Cava. For femoral vein approach, catheter should be advanced into vessel so catheter tip lies parallel to vessel wall and does not enter right atrium. Catheter tip location should be confirmed according to institutional policy and procedure.
4. Clinicians must be aware of potential entrapment of the guidewire by any implanted device in circulatory system. It is recommended that if patient has a circulatory system implant, catheter procedure be done under direct visualization to reduce risk of guidewire entrapment.
5. Do not use excessive force when introducing guidewire or tissue dilator as this can lead to vessel perforation, bleeding, or component damage.
6. Passage of guidewire into the right heart can cause dysrhythmias, right bundle branch block, and a perforation of vessel, atrial or ventricular wall.
7. Do not apply excessive force in placing or removing catheter or guidewire. Excessive force can cause component damage or breakage. If damage is suspected or withdrawal cannot be easily accomplished, radiographic visualization should beobtained and further consultation requested.
8. These catheters are not indicated for power injection. The use of catheters not rated for power injection in high-pressure contrast delivery may cause inter-lumen crossover, rupture, or structural failure, posing a risk of patient harm.
9. Do not secure, staple and/or suture directly to outside diameter of catheter body or extension lines to reduce risk of cutting or damaging the catheter or impeding catheter flow. Secure only at indicated stabilization locations.
10. Air embolism can occur if air is allowed to enter a vascular access device or vein. Do not leave open needles or uncapped, unclamped catheters in central venous puncture site. Use only securely tightened Luer-Lock connections with any vascular access device to guard against inadvertent disconnection.
11. Watch out for any early signs of potential complications, such as infection, embolism or phlebitis. Treatment needs when any of the signs occurs.
12. Before dialysis, recommend extracting the fluid in the catheter lumen to prevent the systemic syndrome from heparin.
13. After heparin extracted (aspirated completely) the catheter should be flushed with Sterile saline.
14. In case of thrombus in the lumen, rinse by force is NOT Allowed, try suck the clog by syringe firstly, if it doesn’t work, use blood clot dissolvent.
Note: Only experienced physician is allowed.
15. In case of the flowrate of the catheter is insufficient, consider the following procedures: - Adjust the location of catheter tip according to the specific vascular.
- Connect artery and vena to increase blood flow.
- Refer to the procedure of Item 14 if caused by thrombus.
- To reduce any risk from incompatibility of other devices, replacement of accessories with other devices is prohibited.
16. Please avoid sharp tools touching the extension tube.
17. It is prohibited to use haemostatic forceps to clamp the extension tubes and hubs.
Contraindications
1. Hypersensitivity to device materials.
2. Infection or cut wound around the puncture area.
3. Dysfunction of blood coagulation.
4. During the anticoagulant treatment.
5. Symptoms of inadaptability to puncture operation, such as Pneumothorax, vein sclerosis.
6.Abnormal or unclear anatomical situation at the penetration area, such as sever emphysema, obviously inadaptability from previous operation.
Complications
The clinical benefit of the use of HC’s must be evaluated against the recognized risks and complications of the procedure which include but are not limited to:
- Bleeding
- Arterial puncture
- Arrhythmia
- Air embolism
- Pneumothorax or hemothorax
- Catheter-related bloodstream infection (CRBSI)
- Thoracic duct injury (with left subclavian or left internal jugular approach)
- Venous thrombosis, pulmonary emboli
- Venous stenosis
- Catheter embolization
|
Number of Lumen |
O.D. |
Effective Length/cm |
|
|
Fr |
mm |
||
|
Single |
7Fr |
2.40 |
10, 13, 13.5, 15, 16, 17.5, 20, 24, 25 |
|
8Fr |
2.70 |
10, 13, 13.5, 15, 16, 17.5, 20, 24, 25 |
|
|
11.5F |
3.80 |
13, 15, 16, 20, 24, 25 |
|
|
Double |
6.5Fr |
2.20 |
5, 8, 10, 13, 15, 16, 17.5, 20, 24, 25 |
|
7Fr |
2.40 |
8, 10, 12, 13, 15, 16, 20, 24, 25 |
|
|
8Fr |
2.70 |
10, 13,15, 16, 20, 24, 25 |
|
|
8.5F |
2.85 |
5, 8, 10, 11, 12, 13, 15, 16,
17, 17.5, 20, 24, 25
|
|
|
9Fr |
3.00 |
10, 12, 15, 17, 20, 24, 25 |
|
|
10Fr |
3.40 |
8, 10, 12, 13, 15, 16, 17, 20, 24, 25 |
|
|
11.5F |
3.80 |
10, 12, 13, 13.5, 15, 16, 17.5, 19, 20, 24, 25 |
|
|
12Fr |
4.00/ |
10, 12,13, 13.5, 15, 16, 18, 19, 20, 24, 25 |
|
|
13.5F |
4.50 |
10, 13, 15, 16, 20, 24, 25 |
|
|
14Fr |
4.70 |
10, 12,13, 15, 16, 19, 20, 24, 25 |
|
|
Triple |
11.5F |
3.80 |
10, 13, 15, 16, 19, 20, 24, 25 |
|
12Fr |
4.00 |
10, 13, 15, 16, 19, 20, 24, 25 |
|
Notes:
1) The multi-lumen variants provide dedicated lumens for haemodialysis therapy, infusion therapy, pressure monitoring.
2) The specification of haemodialysis catheter can be selected based on the patient's age and medical condition.
3) OD of 6.5Fr, 7 Fr, 8Fr and 8.5Fr are intended for pediatric use.
4) Use of this product should be restricted to a trained healthcare professional.
For more information, please contact your sales representative or call +86-757-89959515.
Guangdong Provincial Medical Device Approval No. [2025]007127